Organic chemistry is a subdiscipline of chemistry that studies the structure, properties and reactions of organic compounds, which contain carbon in covalent bonding.
Read More On
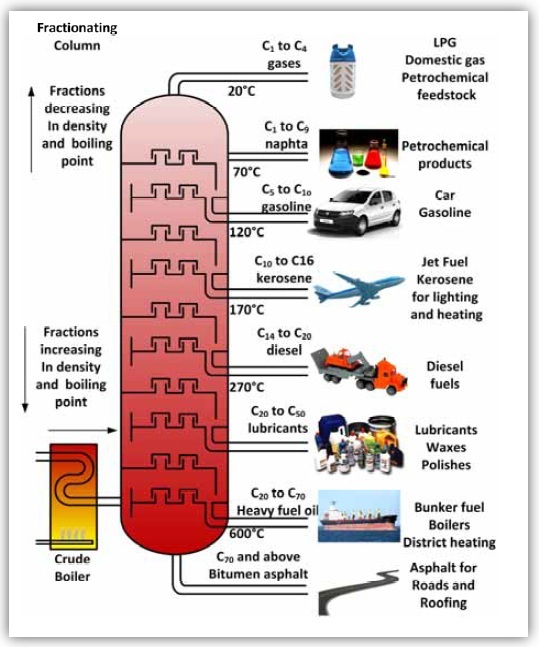
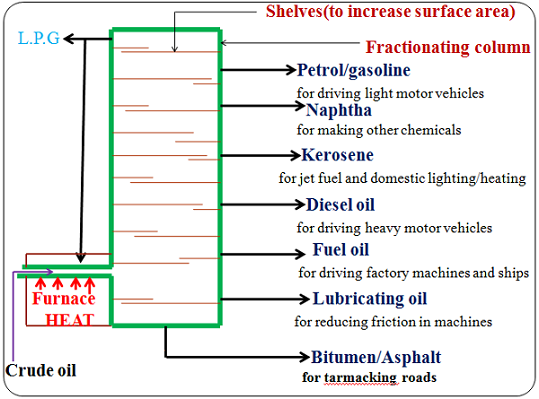

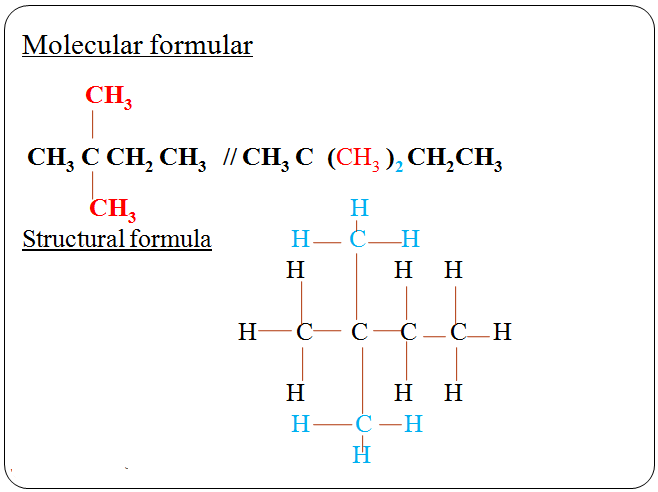
CH4 (g) + 2O2(g) → CO2(g) + 2H2O(g)
The general reaction is as followsCnH2n+n (g) + (1.5n+0.5)O2(g) → nCO2(g) + (n+1)H2O(g)
If there is not enough oxygen present then instead of carbon dioxide, carbon monoxide, CO, is produced.